Key Concepts for Medical Device Entrepreneurs
Lessons Learned from Simbex’s Federally-Funded Medical Device Commercialization Centers
At Simbex, we work with healthcare startups, established medical companies, medtech entrepreneurs, and other innovators to help bring their device concepts to life. We’ve been designing and developing medical devices and health and wellness products for clients, as well as bringing our own innovations to market, for over twenty years!
Our holistic approach to product design centers on identifying risk at every stage of the commercialization process from concept to market and planning ahead to lower the level of risk as much as possible. We never say “We’ll worry about that when we get there!”
What you might not know about Simbex is that we’ve honed this approach over a decade of running innovation commercialization programs for the National Institutes of Health and the Food and Drug Administration. Both NIH and FDA recognized the importance of helping medical device entrepreneurs navigate the commercialization pathway, particularly the early market assessment and business model development milestones that are often overlooked or underestimated for their importance.
They awarded grants to Simbex to help guide innovators in the rehabilitation and assistive technology space through our NIH-funded Center for Translation of Rehabilitation Engineering Advances and Technology (TREAT) and in the pediatric medical device space through the FDA-funded New England Pediatric Device Consortium (NEPDC). Simbex continues to advise biomedical entrepreneurs through the NIH-funded DRIVEN Accelerator Hub.
Simbex Has Provided Assessment & Advisement on More Than 900 Medical Device Concepts
Through these federally supported consortia, grant funds were thoughtfully allocated to develop impactful programming, providing education, project assessment, and financial as well as in-kind assistance to reach as many innovators as possible.
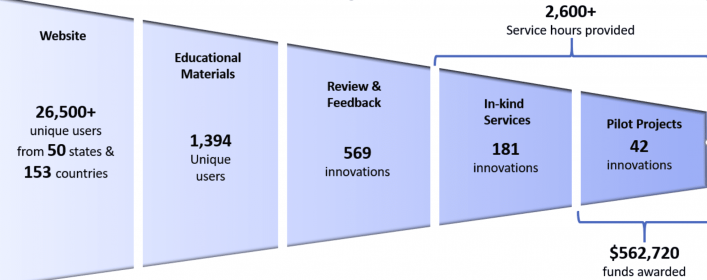
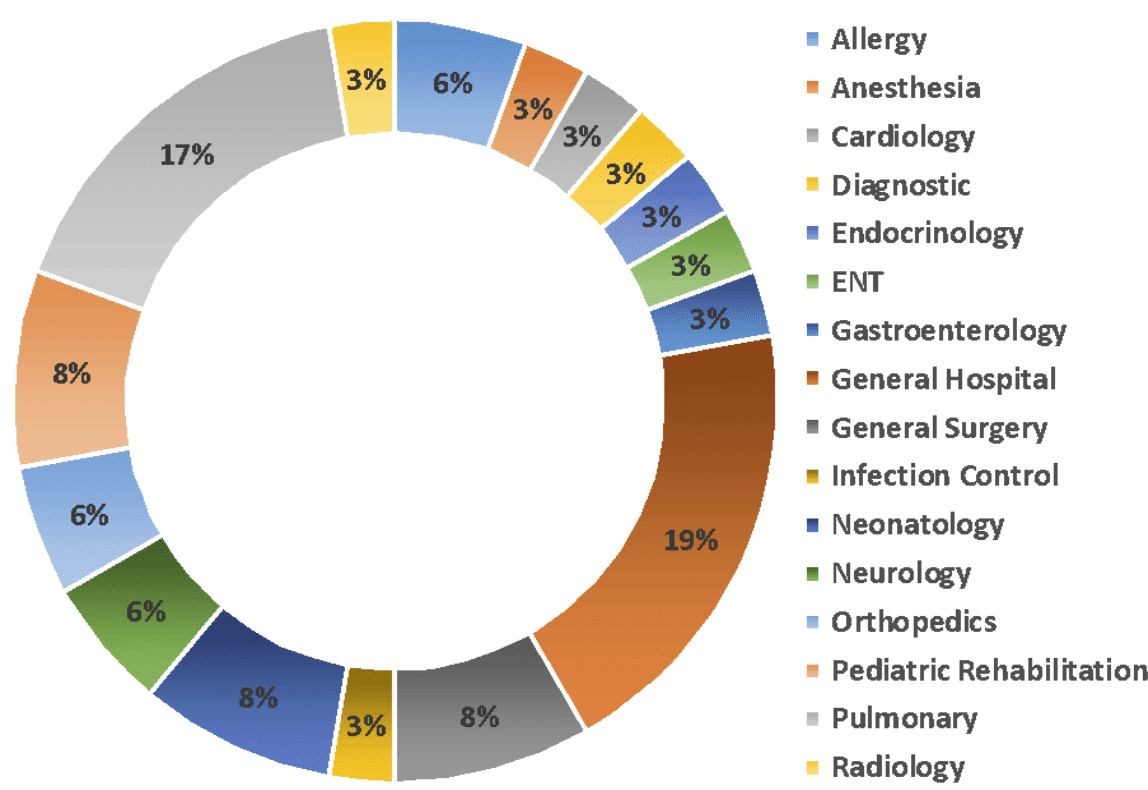
Across all 900 device concepts, the same challenges were presented to us again and again by the innovators who engaged with our commercialization centers, reinforcing the need for the programs and guidance we provided. Below are some of the most common topic areas we covered, which remain at the heart of the commercialization consulting services we provide today.



