
NeuroCool is a portable, non-invasive, selective brain-cooling device that provides emergency neuroprotection by cooling the neck, without the need for sedation or complications associated with whole-body cooling.

I was a clinician-scientist with a dream and data. But I lacked the skills to convert that into a product, which is why I came to Simbex. They had all of the engineering, regulatory, and commercialization experience I needed, all in one place.
Dr. Adam Sprouse-Blum, Founder
Cardiac Arrest: Cool the Brain
Background:
During cardiac arrest, blood flow to the brain ceases, leading to death if not intervened upon quickly. Only 26% of patients survive the initial insult. For those who do, the American Heart Association recommends brain cooling to improve neurologic outcomes. Brain cooling works by lowering metabolic activity, inhibiting the release of harmful neurochemicals, and maintaining the blood-brain barrier.
Post-cardiac arrest brain injury is the main cause of death in patients resuscitated from cardiac arrest, and the main cause of long-term disability in survivors.
Therapeutic hypothermia, or brain cooling, is the most effective neuroprotective intervention for post-cardiac arrest brain injury. It works by rapidly reducing cerebral metabolic rate, suppressing excitotoxic neurotransmitter release, and stabilizing the blood-brain barrier. Immediate initiation following cardiac arrest is critical, as studies show that early cooling significantly improves neurologic outcomes.
The Two-and-a-Half-Hour Delay
Current brain cooling methods are both slow and imprecise. Today, treatment relies on whole-body cooling, which requires sedation and is typically limited to the ICU setting. This requirement causes a critical delay in care and limits when and where treatment can begin.
On average, brain cooling does not start until more than 2.5 hours after patient revival. For patients in rural areas, delays can be even longer due to transport time and reduced access to ICU-level care. These delays have measurable consequences, as earlier intervention significantly improves outcomes.
When brain cooling is initiated within 2 hours of hospital arrival, survival rates increase to 52%, compared to 40% when treatment is delayed. Despite the immediacy, current approaches make it difficult to deliver timely, focused interventions when they matter most.
From Hypothesis to Solution
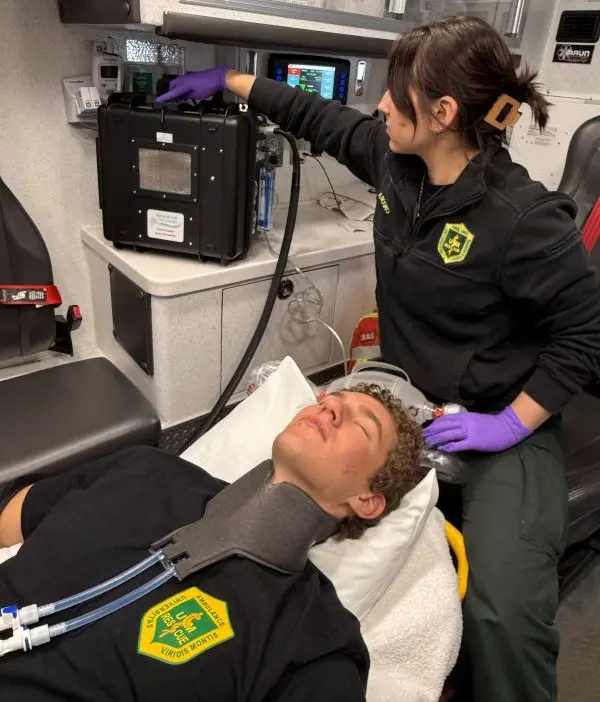
Dr. Sprouse-Blum, a University of Vermont physician, identified a significant gap in emergency neuroprotection. Existing methods rely on ICU-based whole-body cooling, delaying intervention when time is critical. He saw the need for a portable solution to start cooling immediately—ideally in the ambulance—bringing treatment directly to the patient.
Dr. Sprouse-Blum then focused on a key physiological insight: about 72% of the brain’s blood flows through the carotid arteries in the anterior neck. He hypothesized that cooling this region could reduce brain temperature without affecting the rest of the body. Early testing in healthy volunteers confirmed the concept, showing that localized neck cooling measurably lowered brain temperature.
After a colleague’s recommendation, Dr. Sprouse-Blum partnered with Simbex to create a practical, deployable solution. This collaboration produced NeuroCool—a portable device for early brain cooling, even in pre-hospital settings. By circulating cold fluid through an adhesive neck wrap, NeuroCool cools the carotid arteries directly. The approach moves from systemic to focal brain cooling, avoids sedation, and closes the 2.5-hour care delay.
How Simbex Met the Challenge
The Simbex team first worked with Dr. Sprouse-Blum to define the product scope and build a structured requirements framework. This is a standard process in medical device development that kept the project grounded and measurable throughout. All work was conducted under Simbex’s ISO 13485 certified Quality Management System.
Geoff Clark overdelivered and achieved all must-haves, all should-haves, and almost all nice-to-haves. Our first-generation prototype exceeded my expectations.
Dr. Adam Sprouse-Blum
Geoff Clark, Senior Systems Engineer, and Yuan Shi, Product Development Engineer, worked together to tackle five key challenges:
MRI Compatibility
Because brain imaging is often part of the post-cardiac-arrest protocol, the patient-worn neck wrap must be safe and functional in an MRI environment. Simbex achieved this via careful material selection and design choices that eliminate magnetic interference without impairing instrument performance.
Dual-Power Architecture
Simbex engineered an efficient dual-power architecture that can run on battery or be plugged in. It meets the cooling unit’s power requirements while keeping within size and weight constraints, making it practical for EMS personnel to carry and use.
Surrogate Model
Simbex developed a surrogate model to simulate the human body, performed rigorous bench-top testing, and generated a comprehensive testing report.
Component Optimization
To simplify the development process, Simbex identified, modified, and integrated off-the-shelf components to meet performance, reliability, and cost targets. This approach accelerated the development timeline without jeopardizing quality, an important consideration for a grant-funded project.
Ambulance-Ready Design
The device was designed for actual emergency use: intuitive for EMTs to operate quickly within the tight space constraints and workflow of an ambulance.
The Result
The NeuroCool first-generation prototype lowered neck skin temperature to nearly 0°C, exceeding second-generation targets. This enables development to focus on system optimization and clinical integration, accelerating progress toward clinical trials.
Building on this technical achievement, Simbex supported Dr. Sprouse-Blum by actively assisting with the grant application process, providing guidance on engineering, regulatory affairs, and commercialization strategies. Simbex’s support letters helped Dr. Sprouse-Blum demonstrate both technical depth and credibility in his submissions.
Having Simbex as a partner has really bolstered the quality of my grant applications because I’m able to show deep expertise in all of the areas needed to bring a medical device to market.
Dr. Adam Sprouse-Blum
A Partnership Built on More Than Engineering
What stands out to Dr. Sprouse-Blum isn’t just the technical output. Early in the project, Dr. Sprouse-Blum told Greg Lange, CEO of Simbex, that he wanted to learn the medical device development process, not just receive a deliverable. Greg quickly connected him with a funded online educational curriculum and arranged meetings with Simbex and the University of Vermont engineering teams.
This collaborative spirit extended beyond formal arrangements. For example, during one visit, Dr. Sprouse-Blum mentioned a new problem they had discovered with Bluetooth transmission, which was outside the project’s scope. A Simbex engineer, whom Dr. Sprouse-Blum hadn’t met before, immediately offered help and walked Dr. Sprouse-Blum through testing with a Bluetooth dongle.
There was this sense of collaboration that we were all in it together, enjoying the process of solving problems and working towards a common goal… These are just good people out to create medical devices that help humanity.
Dr. Adam Sprouse-Blum




